- Oct 25, 2015
- 5,776
- Pool Size
- 28000
- Surface
- Plaster
- Chlorine
- Salt Water Generator
- SWG Type
- CircuPool RJ-60 Plus
Folks,
I've been really busy with fine tuning the pool recently. We're going on vacation for 18 days (record for us). We have pool automation set up to use esp32 DIY controls... not anywhere as pretty and configurable as the Raspberry Pi solution others on here are using but I just don't have the time to work on that solution for at least a few months. I hope to then restart the Node JS project that many have been so helpful with especially @Katodude, @MyAZPool, and the guy that wrote most of the code who is also a member here (can't remember his id). In the meantime, my esp32 solution was quick, functional, and dirt cheap. Now I need to figure out how to get pH reasonably controlled while we're gone. I know I could do this with acid injection and even just get a pool service to add about a gallon every 3 days but I'd like to avoid both of those options. At a gallon every 3 days, I will require 3.7 pounds of CO2 every three says or say 1.3 # per day. This calculation makes some simplifying assumptions like no loss from off gasing and one mole of CO2 is ultimately equivalent to on mole of HCL. I can get a 50 # cylinder and that should last an entire month at least. I'm assuming in this calculation I get 100% conversion to carbonic acid. So here's a couple questions for the experts:
Chris
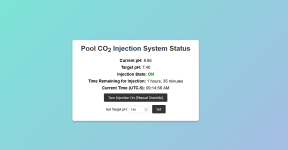
I've been really busy with fine tuning the pool recently. We're going on vacation for 18 days (record for us). We have pool automation set up to use esp32 DIY controls... not anywhere as pretty and configurable as the Raspberry Pi solution others on here are using but I just don't have the time to work on that solution for at least a few months. I hope to then restart the Node JS project that many have been so helpful with especially @Katodude, @MyAZPool, and the guy that wrote most of the code who is also a member here (can't remember his id). In the meantime, my esp32 solution was quick, functional, and dirt cheap. Now I need to figure out how to get pH reasonably controlled while we're gone. I know I could do this with acid injection and even just get a pool service to add about a gallon every 3 days but I'd like to avoid both of those options. At a gallon every 3 days, I will require 3.7 pounds of CO2 every three says or say 1.3 # per day. This calculation makes some simplifying assumptions like no loss from off gasing and one mole of CO2 is ultimately equivalent to on mole of HCL. I can get a 50 # cylinder and that should last an entire month at least. I'm assuming in this calculation I get 100% conversion to carbonic acid. So here's a couple questions for the experts:
- Is 100% conversion reasonable to assume? @JoyfulNoise @JamesW any comments/suggestions
- from you on this?
- Seems like I'd be better off injecting down stream of my swg so I don't have too low pH that might remove catalyst on my swg generator, pump seals etc.. That's the last thing in the flow at the equipment pad. Does this make sense to you?
- Control should be pretty simple is there anything I'm missing on the web page below?
Chris

Last edited:








